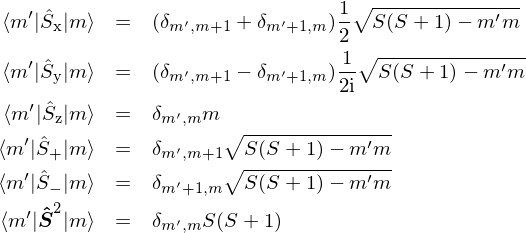Calculate the emf of the following cell at 298 K ? Fe(s)|Fe^2 + (0.001 M )||H^+(1M)|H2(g) (1 bar), Pt(s) (Given: E^∘cell = + 0.44 V )

Wavelength and Frequency E = h c = c = speed of light (3 x 10 8 m/s) = frequency (s -1 ) = wavelength (m) E = energy (Joules or J) h = Planck's constant. - ppt download